Laboratory of Pharmacokinetic Optimization
1. Members
| Adjunct Project Leader | NAGATOISHI Satoru |
|---|
2. Background and objectives
We research on creating new types of biopharmaceuticals through the search for seeds of biopharmaceuticals, including antibodies, and research on optimization technologies for these seeds. We aim to obtain functional molecules with excellent physical properties and reliably evaluate their performance by characterizing antibodies obtained from various animal species using various analytical techniques, amino acid mutation, molecular fusion, antibody modification by conjugation, and solution property control using solvent science. In addition to antibodies, we develop fundamental technologies and provide optimum support for a wide variety of pharmaceuticals focusing on drug discovery modalities.
3. Overview of our research
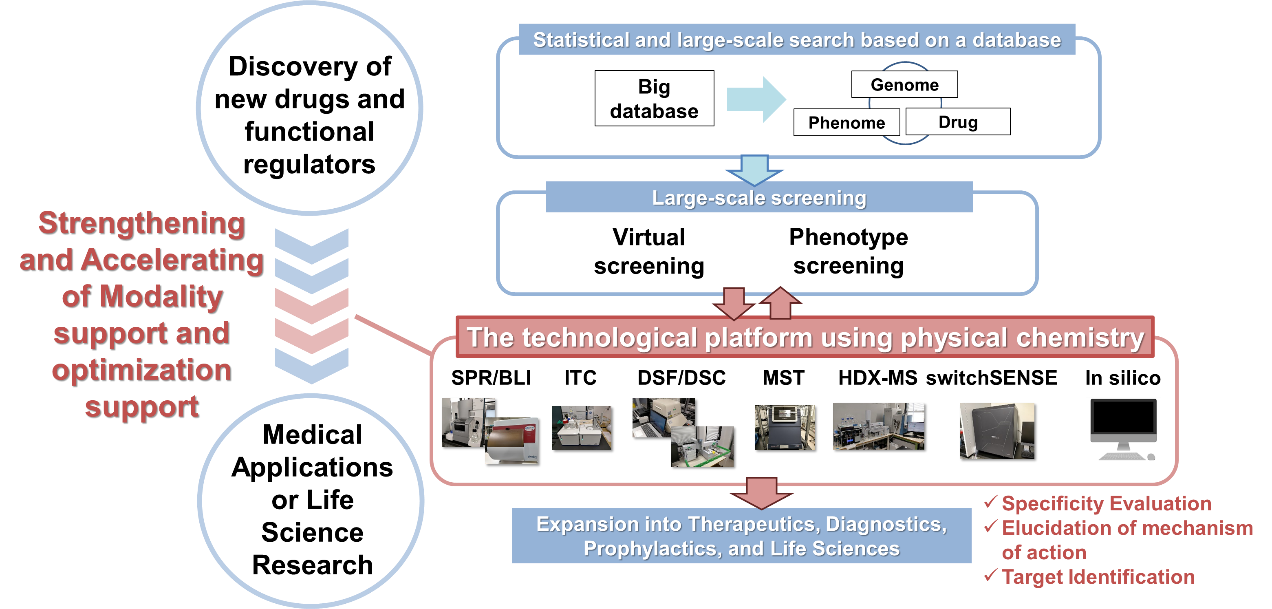
We will clarify the functions and properties required for next-generation biopharmaceuticals in particular by analyzing the physical properties of the small, medium, and macromolecular drug discovery modalities, focusing on antibodies. We also conduct basic and practical research on biomolecular modification technologies to support optimization.
3-1.Basic research on antibody modalities
Specific and strong affinity is required for the production of targeting ligands such as antibodies. We are studying methods to improve the affinity of ligands by utilizing the affinity generation mechanism understood from the above-mentioned research. Specifically, in addition to the acquisition of molecules by phage display technology, we are working to improve the physical properties of various types of antibodies, including their affinities, by combining different antibody species, conjugation of functional molecules, and computational approaches.
Reference:
Watanabe Y, Tanabe A, Hamakubo T, Nagatoishi S, Tsumoto K.: Development of biparatopic bispecific antibody possessing tetravalent scFv-Fc capable of binding to ROBO1 expressed in hepatocellular carcinoma cells., J Biochem. 170(2):307-315, 2021.
Yoshida K, Kuroda D, Kiyoshi M, Nakakido M, Nagatoishi S, Soga S, Shirai H, Tsumoto K.: Exploring designability of electrostatic complementarity at an antigen-antibody interface directed by mutagenesis, biophysical analysis, and molecular dynamics simulations, Sci Rep. 9(1):4482, 2019.
Yamashita T, Mizohata E, Nagatoishi S, Watanabe T, Nakakido M, Iwanari H, Mochizuki Y, Nakayama T, Kado Y, Yokota Y, Matsumura H, Kawamura T, Kodama T, Hamakubo T, Inoue T, Fujitani H, Tsumoto K.: Affinity Improvement of a Cancer-Targeted Antibody through Alanine-Induced Adjustment of Antigen-Antibody Interface, Structure. 27(3):519-527.e5, 2019.
Egashira Y, Nagatoishi S, Kiyoshi M, Ishii-Watabe A, Tsumoto K.: Characterization of glycoengineered anti-HER2 monoclonal antibodies produced by using a silkworm-baculovirus expression system, J Biochem, 163(6):481-488, 2018.
Yui A, Akiba H, Kudo S, Nakakido M, Nagatoishi S, Tsumoto K.: Thermodynamic analyses of amino acid residues at the interface of an antibody B2212A and its antigen roundabout homolog 1, J Biochem, 162(4):255-258, 2017.
Yumura K, Akiba H, Nagatoishi S, Kusano-Arai O, Iwanari H, Hamakubo T, Tsumoto K.: Use of SpyTag/SpyCatcher to construct bispecific antibodies that target two epitopes of a single antigen, J Biochem, 162(3):203-210, 2017.
Kiyoshi M, Caaveiro JM, Miura E, Nagatoishi S, Nakakido M, Soga S, Shirai H, Kawabata S, Tsumoto K.: Affinity improvement of a therapeutic antibody by structure-based computational design: generation of electrostatic interactions in the transition state stabilizes the antibody-antigen complex, PLoS One, 9(1):e87099, 2014.
3-2.Basic research on drug discovery modalities
We research on the discovery and optimization of small, medium, and macromolecular ligands that regulate biomolecular interactions. We also develop and propose new discovery techniques by introducing methods that enable precise knowledge of the physical properties of biomolecules and precise analysis of their functions and interactions. We utilize physicochemical analysis methods (ITC, SPR, DSC, MST, HDX-MS, etc.) in exploration (screening), selection (validation), and optimization (hit-to-lead, optimization) to determine the "quality" of interactions without being limited by binding affinity. We are also attempting to obtain novel ligands for challenging target molecules by analyzing the "ligand" quantitatively. In close collaboration with the DDI of the University of Tokyo and BINDS, a drug discovery support project of AMED, we aim to apply this technology to therapeutic and diagnostic drugs.
Reference:
Senoo A, Ito S, Nagatoishi S, Saito Y, Ueno G, Kuroda D, Yoshida K, Tashima T, Kudo S, Sando S, Tsumoto K.: Regulation of cadherin dimerization by chemical fragments as a trigger to inhibit cell adhesion., Commun Biol. 4(1):1041, 2021.
Yoshimura C, Nagatoishi S, Kuroda D, Kodama Y, Uno T, Kitade M, Chong-Takata K,: Thermodynamic Dissection of Potency and Selectivity of Cytosolic Hsp90 Inhibitors, J Med Chem. 64(5):2669-2677, 2021.
Nagatoishi S, Caaveiro JMM, Tsumoto K.: Biophysical Analysis of the Protein-Small Molecule Interactions to Develop Small Molecule Drug Discovery, Yakugaku Zasshi, 138(8):1033-1041, 2018.
Tashiro S, Caaveiro JMM, Nakakido M, Tanabe A, Nagatoishi S, Tamura Y, Matsuda N, Liu D, Hoang QQ, Tsumoto K.: Discovery and Optimization of Inhibitors of the Parkinson's Disease Associated Protein DJ-1, ACS Chem Biol. 13(9):2783-2793, 2018.
Senoo A , Nagatoishi S , Moberg A , Babol LN , Mitani T , Tashima T , Kudo S , Tsumoto K.: Inhibition of homophilic dimerization and disruption of cell adhesion by P-cadherin-specific small molecules from SPR-based assays, Chem Commun, 54(42):5350-5353, 2018.
Nagatoishi S, Yamaguchi S, Katoh E, Kajita K, Yokotagawa T, Kanai S, Furuya T, Tsumoto K.: A combination of 19F NMR and surface plasmon resonance for site-specific hit selection and validation of fragment molecules that bind to the ATP-binding site of a kinase, Bioorg Med Chem, 26(8):1929-1938, 2018.
Nakano K, Chigira T, Miyafusa T, Nagatoishi S, Caaveiro JM, Tsumoto K.: Discovery and characterization of natural tropolones as inhibitors of the antibacterial target CapF from Staphylococcus aureus, Sci Rep, 5:15337, 2015.
3-3.Basic research on functional control by physicochemical analysis techniques in pharmaceuticals
Biomolecular interactions and drug interactions are based on specific molecular recognition that skillfully utilizes hydrogen bonding and van der Waals forces. We are studying the molecular recognition by proteins, especially antibodies, and how the specific interactions are established through two aspects: structural biology and physical chemistry. Through these studies, we are trying to clarify the role of each functional group of biomolecules and elucidate the mechanism of the precise recognition function of these molecules. Based on the findings, we are also developing guidelines and new technologies for drug design.
Reference:
Ten T, Nagatoishi S, Maeda R, Hoshino M, Nakayama Y, Seiki M, Sakamoto T, Tsumoto K.: Structural and thermodynamical insights into the binding and inhibition of FIH-1 by the N-terminal disordered region of Mint3, J Biol Chem, 297(5):101304, 2021.
Yui A, Caaveiro JMM, Kuroda D, Nakakido M, Nagatoishi S, Goda S, Maruno T, Uchiyama S, Tsumoto K.: Mechanism of dimerization and structural features of human LI-cadherin, J Biol Chem, 297(3):101054, 2021.
Yoshida K, Nagatoishi S, Kuroda D, Suzuki N, Murata T, Tsumoto K.: Phospholipid Membrane Fluidity Alters Ligand Binding Activity of a G Protein-Coupled Receptor by Shifting the Conformational Equilibrium, Biochemistry. 58(6):504-508, 2019.
Tashima T, Nagatoishi S, Caaveiro JMM, Nakakido M, Sagara H, Kusano-Arai O, Iwanari H, Mimuro H, Hamakubo T, Ohnuma SI, Tsumoto K.: Molecular basis for governing the morphology of type-I collagen fibrils by Osteomodulin, Commun Biol. 1:33, 2018.
Kudo S, Caaveiro JM, Nagatoishi S, Miyafusa T, Matsuura T, Sudou Y, Tsumoto K.: Disruption of cell adhesion by an antibody targeting the cell-adhesive intermediate (X-dimer) of human P-cadherin, Sci Rep, 7:39518, 2017.
Ota C, Noguchi S, Nagatoishi S, Tsumoto K.: Assessment of the Protein-Protein Interactions in a Highly Concentrated Antibody Solution by Using Raman Spectroscopy, Pharm Res, 33(4):956-69, 2016.
Nakayama T, Mizohata E, Yamashita T, Nagatoishi S, Nakakido M, Iwanari H, Mochizuki Y, Kado Y, Yokota Y, Satoh R, Tsumoto K, Fujitani H, Kodama T, Hamakubo T, Inoue T.: Structural features of interfacial tyrosine residue in ROBO1 fibronectin domain-antibody complex: Crystallographic, thermodynamic, and molecular dynamic analyses, Protein Sci, 24(3):328-40, 2015.
Laboratory of Pharmacokinetic Optimization
| Website | http://park.itc.u-tokyo.ac.jp/phys-biochem/ |
|---|---|
| satoru-nagatoishi※nibiohn.go.jp (Please replace ※ with @.) |
