Laboratory of Proteome Research
1. Members
| Project leader | ADACHI Jun |
|---|---|
| Senior Research fellow | TOMONAGA Takeshi |
| Senior Researcher | MURAOKA Satoshi |
| Postdoctoral associate | NARUMI Ryohei, SATO Yumi, ITO Hisahiro |
| Researcher | ISOYAMA Junko |
| Technical assistant | ISHIDA Mimiko, IKEMOTO Narumi, HIRANO Masayo, TAKADA Yoko, KASUYA Yoko |
| Collaborative Research Scientist | ABE Yuichi, HIRANO Hidekazu, KUGA Takahisa, MATSUBARA Misako, HARA Yasuhiro, SHIROMIZU Takashi |
| Student | GUNJI Daigo |
| Administrative Assistant | HYODO Seiko |
2. Background and Aims
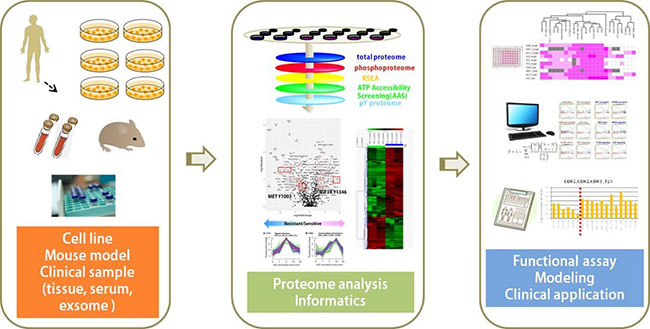
Many human diseases are caused by aberrant function of proteins. Therefore, it is obvious that development of novel biomarkers useful for diagnosis and treatment of the diseases needs to find such abnormal proteins. Proteomics is a powerful method to comprehensively analyze such proteins. Recent advances in proteomic technology made it possible to identify disease-related proteins in the clinical samples and thus extensive efforts are now attempted to search for the biomarkers across the country, however, none of them has been accepted for clinical use. The main reason is that candidate proteins for biomarker have not been evaluated if they are really involved in the development and progression of the diseases. In this project, our goal is to identify bona fide biomarkers useful for diagnosis and treatment of human diseases through functional analysis of candidate proteins by combining most recent proteomic technology with molecular and cellular biology.
3. Overview of our research
I. Identification of novel biomarker candidate proteins and peptides
Our project develops and applies methods of MS-based proteomics especially for clinical proteomics. Using patient-derived cell lines, mouse models and clinical samples, we aimed to identify biomarkers whose expressions alter quantitatively and qualitatively along with the development and progression of diseases. Serum and plasma samples and exsomes in these samples are used for the analysis of biomarkers useful for diagnosis, while tissue samples from various lesions are used for identification of target proteins for drug discovery.
II. Evaluation of the candidate proteins and peptides for clinical utility
These biomarker candidate proteins should be validated if they are clinically valuable. Diagnostic utility of the proteins will be evaluated by multicenter clinical trial if they are superior to currently-available biomarkers by themselves or by combination with these widely-used markers. Therapeutic benefit should be evaluated if the function of the proteins participates in the development of diseases. It can be tested by examining the disease-related phenotypes after overexpression or suppression of the candidate proteins in cultured cells or genetically-engineered mice. Validation is also performed using recently developed MRM method with a triple quadrupole mass spectrometer.
Laboratory of Proteome Research
| tomonaga*nibiohn.go.jp (replace * by @) |
